Aspartame
Aspartame is an artificial sweetener, also known as E951. Artificial sweeteners are all substances that do not belong to the group of natural sugars, but which, like sugar, also trigger the sense of sweet taste. Artificial sweeteners commonly serve as a substitute for sugar in many of the products that are sold today.
Aspartame belongs to the group of so-called intense sweeteners. This means that the sweetening power of aspartame is 150 to 200 times greater than sugar, so far less needs to be added. Aspartame hardly releases/ generates any energy. In terms of flavour aspartame comes closest to match sugar’s taste profile compared to other artificial sweeteners. Aspartame also works as a flavour enhancer for other sweeteners, fruits and fruit aromas, and it does not leave an unpleasant aftertaste.
Aspartame is the chemical combination of the amino acids aspartic acid and L-phenylalanine, with at the phenylalanine-part a methanol group. This particular combination does occur naturally, however, the methyl ester makes that this substance is synthetic. Due to the phenylalanine, a warning stating 'Contains a source of phenylalanine' must always be placed on the packaging of a product containing aspartame.
In acidic products aspartame is very stable, but when aspartame is heated it will disintegrate and lose its sweet taste. Aspartame is therefore mainly used in diet sodas and other foods that are prepared cold.
The production process of aspartame
Fermentation
By using the bacteria Brevibacterium flavum and Corynebacterium glutamicum through direct fermentation, the amino acids L-aspartic acid and L-phenylalanine are formed. The fermentation process starts with a pure culture of bacteria, which will multiply in the presence of the necessary nutrients.
When sufficient bacteria have been formed during the pre-fermentation, the bacteria are transferred to a tank which contains all the nutrients necessary to form more bacteria. In addition, sources of carbon and nitrogen are present, which are necessary for the forming of large quantities of amino acids. This mass is continuously kept in motion with a mixer and with a pump filtered compressed air is added to the mix. In a fermentation tank containing the same nutrients, the bacteria get the chance to produce large quantities of amino acids. The desired pH-value is maintained by using ammonia water.
Isolation
When sufficient amino acids have been formed, the desired amino acids have to be separated from the ones that are not needed. Using a centrifuge, a large quantity of the amino acids is separated from the bacteria. In an ion exchanger the required amino acids are further separated and purified. These amino acids are then pumped into a crystallization tank, after which the crystals that have formed are removed by using a centrifuge. Subsequently, the amino acids are dried and prepared for synthesis to aspartame.
Modification
Usually, the synthesis of aspartame starts with the modification of aspartic acid and phenylalanine.
The aspartic acid is modified in such a way that certain parts of the molecule are shielded, by for example benzene rings, ensuring that further chemical reactions will only take place with the right acid group of the aspartic acid.
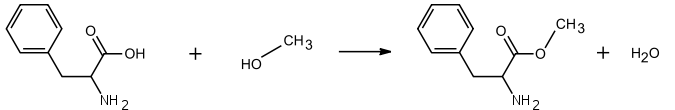
Through a reaction of the phenylalanine with the methanol, L-phenylalanine are created with a methyl ester, as shown in the chemical reaction below.
Synthesis
The modified amino acids are then pumped to a reactor, where they are mixed at room temperature during 24 hours. Next, the temperature is increased to 65°C, and this temperature will be maintained for another 24 hours. After cooling the mixture down to minus 18°C, a solvent is added to dilute the mixture, so crystallization can take place. The crystals which are formed during this process are isolated by filtration and dried.
Modification
In a large tank, the crystals that were formed are converted to aspartame through a reaction with acetic acid. In this tank the crystals, acetic acid solution, palladium catalyst and hydrogen are mixed together intensely for about 12 hours, causing them to chemically react with each other.
Purification
After the 12 hours of chemical reactions have passed, the palladium catalyst is removed by filtration and the solvent is removed through distillation. The remaining solid residue is dissolved with ethanol and after that recrystallized. The aspartame crystals that were produced during this process are filtered and dried.
Food Safety & Hygienic Design
The fermentation process creates the ideal conditions for micro-organisms to grow. Especially unwanted micro-organisms. The part of the production process where the fermentation takes place should be designed aseptically. This means that the machinery and equipment should be cleanable to a microbial level, steam- sterilizable and bacteria tight. In many fermentation processes, equipment used is often only steam-sterilizable, and thus not fully cleanable. This can cause the fermentations to give incorrect results. The sterilization of a dirty tank containing shielded micro-organisms, will always be an unreliable factor during sterilization.
The downstream process must be carried out hygienically – and cleanable to a microbial level. Recrystallization is step to purify the product, but it cannot guarantee that microbial contamination is not included by accident. Because the product is not heat stable, it cannot be exposed to heat.
Poor design of an aspartame plant or factory can cause an unexpectedly high germination rate.